|
Narsoplimab, a mannan-binding lectin-associated protease-2 inhibitor, for the treatment of adult hematopoietic stem-cell transplantation–associated thrombotic microangiopathy. The 61% response rate and 68% 100-day survival seen with narsoplimab-treated TA-TMA patients in the pivotal clinical trial represent an approximately three-fold improvement when compared to an untreated age- and disease status-matched cohort from a systematic literature review. Narsoplimab is designed to prevent complement-mediated inflammation and endothelial damage without affecting other pathways of innate immunity. Importantly, unlike other complement-targeting drugs on the market or in development, inhibition of MASP-2 by narsoplimab does not interfere with the classical complement pathway, a critical component of the acquired immune response to infection. The lectin pathway is activated primarily by tissue damage or microbial infection. Narsoplimab, also known as OMS721, is an investigational fully human monoclonal antibody targeting mannan-binding lectin-associated serine protease-2 (MASP-2), the effector enzyme of the lectin pathway of the complement system. For more information about Omeros and its programs, visit View source version on businesswire.Stem Cell Transplant-Associated TMA (TA-TMA) Narsoplimab: Our first-in-class monoclonal antibody complement inhibitor OMS906, Omeros’ inhibitor of MASP-3, the key activator of the alternative pathway of complement, is advancing across multiple clinical programs for alternative pathway-related diseases, including paroxysmal nocturnal hemoglobinuria (PNH) and complement 3 (C3) glomerulopathy. Omeros’ long-acting MASP-2 inhibitor OMS1029 is currently in a Phase 1 clinical trial. Narsoplimab is also in multiple late-stage clinical development programs focused on other complement-mediated disorders, including IgA nephropathy, COVID-19, and atypical hemolytic uremic syndrome. Omeros’ lead MASP-2 inhibitor narsoplimab targets the lectin pathway of complement and is the subject of a biologics license application pending before FDA for the treatment of hematopoietic stem cell transplant-associated thrombotic microangiopathy (TA-TMA). Omeros is an innovative biopharmaceutical company committed to discovering, developing and commercializing small-molecule and protein therapeutics for large-market and orphan indications targeting immunologic disorders including complement-mediated diseases, cancers, and addictive and compulsive disorders. The replay access code is 8266699.įor online access to the live or subsequently archived webcast of the conference call, go to Omeros’ website at. 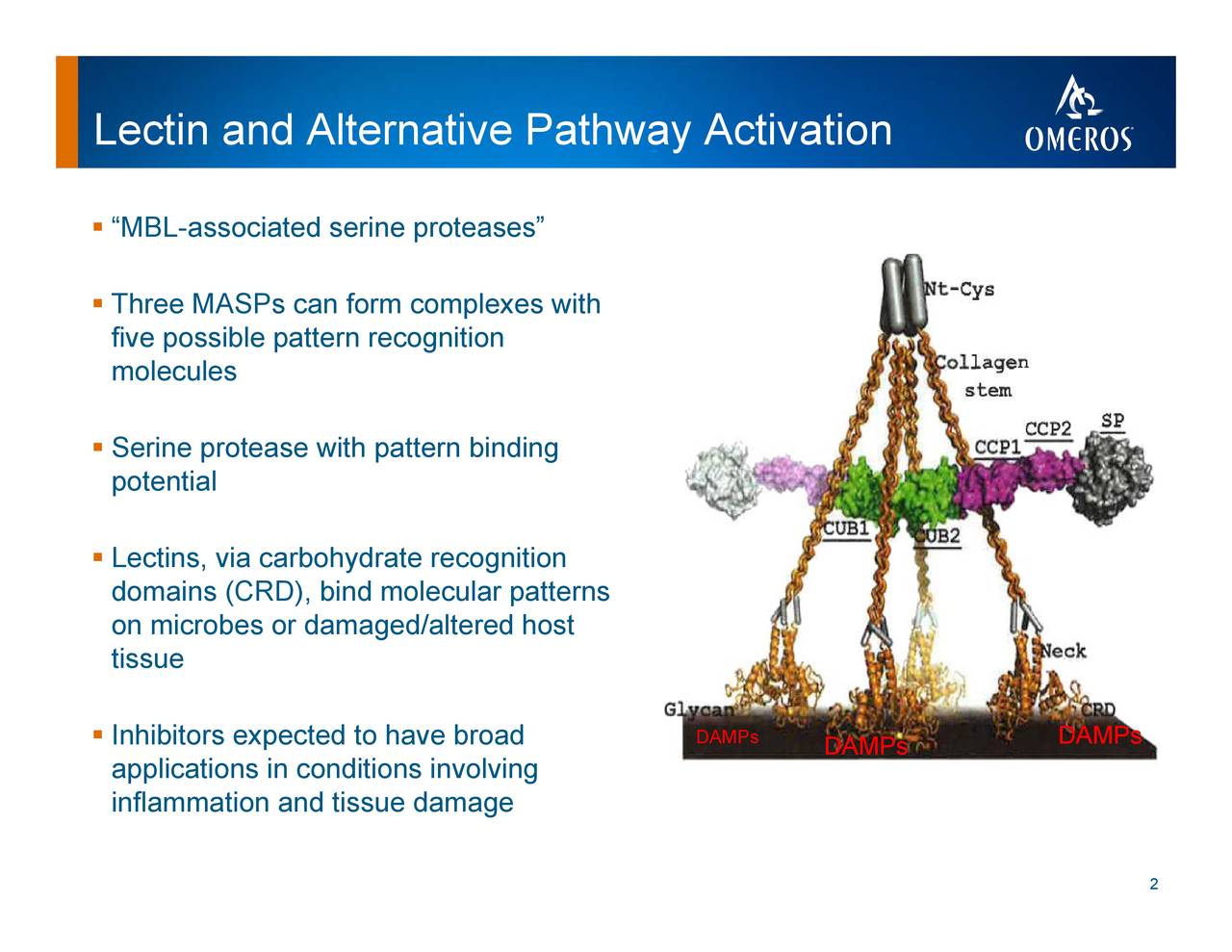
A telephone replay will be available for one week following the call and may be accessed by dialing (800) 770-2030 from the United States or Canada or (609) 800-9909 internationally. Please dial in approximately 10 minutes prior to the start of the call. To access the live conference call via phone, please dial (800) 715-9871 from the United States and Canada or (646) 307-1963 internationally and ask to be placed into the Omeros earnings call.  
Pacific Time) to discuss the financial results as well as recent developments and highlights. Omeros management will host a conference call and webcast that same day at 4:30 p.m. 
SEATTLEOmeros Corporation (NASDAQ: OMER), today announced that the company will issue its first quarter financial results for the period ended March 31, 2023, on Tuesday, May 9, 2023, after the market closes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
